CAS No.: 128-13-2
熊去氧胆酸 Catalog No. CSN12521
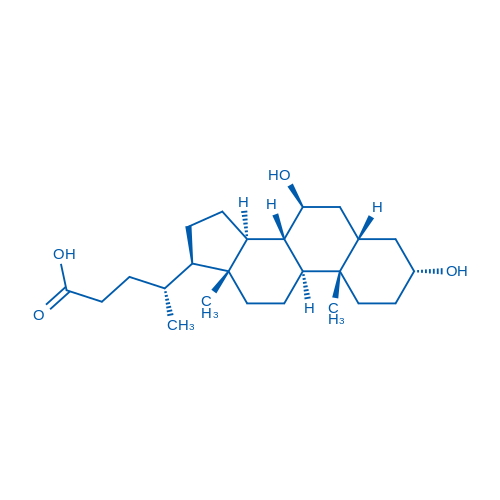
Synonyms: 熊去氧胆酸;Ursodeoxycholate;Ursodiol
Ursodiol showed regulations of immunoregulatory responses via regulation of cytokines, antimicrobial peptides defensins, and take an active part in increased restitution of wound in the colon, as well as anti-inflammatory and protective effects in human epithelial cells of the gastrointestinal tract.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02026401 Primary Biliary Cirrhosis Phase 2 Completed - United States, Arizona ... more >> NGM Clinical Study Site 103 Phoenix, Arizona, United States United States, California NGM Clinical Study Site 108 Coronado, California, United States United States, Michigan NGM Clinical Study Site 101 Detroit, Michigan, United States United States, North Carolina NGM Clinical Study Site 105 Durham, North Carolina, United States United States, Texas NGM Clinical Study Site 102 Dallas, Texas, United States NGM Clinical Study Site 113 San Antonio, Texas, United States United States, Virginia NGM Clinical Study Site 104 Richmond, Virginia, United States Australia, New South Wales NGM Clinical Study Site 602 Sydney, New South Wales, Australia NGM Clinical Study Site 606 Sydney, New South Wales, Australia NGM Clinical Study Site 609 Sydney, New South Wales, Australia NGM Clinical Study Site 611 Sydney, New South Wales, Australia Australia, Queensland NGM Clinical Study Site 614 Brisbane, Queensland, Australia Australia, South Australia NGM Clinical Study Site 607 Adelaide, South Australia, Australia NGM Clinical Study Site 608 Adelaide, South Australia, Australia Australia, Victoria NGM Clinical Study Site 601 Melbourne, Victoria, Australia NGM Clinical Study Site 604 Melbourne, Victoria, Australia NGM Clinical Study Site 613 Melbourne, Victoria, Australia Collapse << NCT00004442 Infantile Refsum's Disease ... more >> Zellweger Syndrome Bifunctional Enzyme Deficiency Adrenoleukodystrophy Collapse << Not Applicable Terminated - - NCT00805805 Primary Biliary Cirrhosis Phase 3 Completed - United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << - 更多
- 参考文献
- [1] Jackson H, Solaymani-Dodaran M, et al. Influence of ursodeoxycholic acid on the mortality and malignancy associated with primary biliary cirrhosis: a population-based cohort study. Hepatology. 2007 Oct;46(4):1131-7.
- [2] Kumar D, Tandon RK. Use of ursodeoxycholic acid in liver diseases. J Gastroenterol Hepatol. 2001 Jan;16(1):3-14.
- [3] Rubin RA, Kowalski TE, Khandelwal M, Malet PF. Ursodiol for hepatobiliary disorders. Ann Intern Med. 1994 Aug 1;121(3):207-18. doi: 10.7326/0003-4819-121-3-199408010-00009. PMID: 8017748.
- [4] Pramar YV, Mandal TK, Bostanian LA, Nguyen AT, Miller V, Morris TC, Graves RA. Stability of Compounded Ursodiol Suspensions in PCCA Base, SuspendIt. Int J Pharm Compd. 2019 Jan-Feb;23(1):70-76. PMID: 30668538.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 128-13-2 | 储存条件 |
|
|||||
| 分子式 | C24H40O4 | 运输 | 蓝冰 | |||||
| 分子量 | 392.57 | 别名 | 熊去氧胆酸;Ursodeoxycholate;Ursodiol;UDCA | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02026401 | Primary Biliary Cirrhosis | Phase 2 | Completed | - | United States, Arizona ... more >> NGM Clinical Study Site 103 Phoenix, Arizona, United States United States, California NGM Clinical Study Site 108 Coronado, California, United States United States, Michigan NGM Clinical Study Site 101 Detroit, Michigan, United States United States, North Carolina NGM Clinical Study Site 105 Durham, North Carolina, United States United States, Texas NGM Clinical Study Site 102 Dallas, Texas, United States NGM Clinical Study Site 113 San Antonio, Texas, United States United States, Virginia NGM Clinical Study Site 104 Richmond, Virginia, United States Australia, New South Wales NGM Clinical Study Site 602 Sydney, New South Wales, Australia NGM Clinical Study Site 606 Sydney, New South Wales, Australia NGM Clinical Study Site 609 Sydney, New South Wales, Australia NGM Clinical Study Site 611 Sydney, New South Wales, Australia Australia, Queensland NGM Clinical Study Site 614 Brisbane, Queensland, Australia Australia, South Australia NGM Clinical Study Site 607 Adelaide, South Australia, Australia NGM Clinical Study Site 608 Adelaide, South Australia, Australia Australia, Victoria NGM Clinical Study Site 601 Melbourne, Victoria, Australia NGM Clinical Study Site 604 Melbourne, Victoria, Australia NGM Clinical Study Site 613 Melbourne, Victoria, Australia Collapse << |
| NCT00004442 | Infantile Refsum's Disease ... more >> Zellweger Syndrome Bifunctional Enzyme Deficiency Adrenoleukodystrophy Collapse << | Not Applicable | Terminated | - | - |
| NCT00805805 | Primary Biliary Cirrhosis | Phase 3 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT03394924 | Primary Biliary Cholangitis | Phase 2 | Recruiting | April 2019 | United States, Arkansas ... more >> Arkansas Diagnostic Center Recruiting Little Rock, Arkansas, United States, 72205 Principal Investigator: Alonzo Williams Texas Clinical Research Institute Recruiting Little Rock, Arkansas, United States, 76012 Principal Investigator: Reem Ghalib United States, California Southern California Research Center Recruiting Coronado, California, United States, 92118 Principal Investigator: Tarek Hassanein Cedars-Sinai Medical Center Recruiting Los Angeles, California, United States, 90048 Principal Investigator: Ayoub Walid California Liver Research Institue Recruiting Pasadena, California, United States, 91105 Principal Investigator: Edward Mena Inland Empire Liver Foundation Recruiting Rialto, California, United States, 92377 Contact: Zeid Kayali, MD Principal Investigator: Zeid Kayali, MD United States, Florida Nature Coast Clinical Research Recruiting Inverness, Florida, United States, 34452 Principal Investigator: Paul Hellstern University of Miami Leonard M. Miller School of Medicine Recruiting Miami, Florida, United States, 33136 Principal Investigator: Cynthia Levy United States, Georgia Consultative Gastroenterology Recruiting Atlanta, Georgia, United States, 30312 Principal Investigator: Gary Richter United States, Maryland Mercy Medical Center-McAuley Plaza Recruiting Baltimore, Maryland, United States, 21202 Contact: Olivia Tang Principal Investigator: Paul Thuluvath Digestive Disease Associates Recruiting Catonsville, Maryland, United States, 21228 Principal Investigator: Natarajan Ravendhran United States, Michigan Henry Ford Hospital Recruiting Detroit, Michigan, United States, 48377 Principal Investigator: Reena Salgia United States, Nebraska CHI Health Recruiting Omaha, Nebraska, United States, 68124 Principal Investigator: Sandeep Mukherjee United States, New York Northwell Health Recruiting Manhasset, New York, United States, 11030 Principal Investigator: David Bernstein Mount Sinai Beth Isreal Recruiting New York, New York, United States, 10016 Principal Investigator: Ira Jacobson Weill Cornell Medical College Recruiting New York, New York, United States, 10024 Principal Investigator: Sonal Kumar United States, Pennsylvania University of Pittsburgh Medical Center - Center for Liver Disease Recruiting Pittsburgh, Pennsylvania, United States, 15213 Principal Investigator: Mordechai Rabinovitz United States, Texas The Liver Institute at Methodist Dallas Medical Center Recruiting Dallas, Texas, United States, 75203 Principal Investigator: Maisha Barnes Liver Consultants of Texas Recruiting Dallas, Texas, United States, 76104 Principal Investigator: Manjushree Gautam Baylor Saint Luke's Medical Center Recruiting Houston, Texas, United States, 77030 Principal Investigator: John Vierling American Research Corporation at the Texas Liver Institute Recruiting San Antonio, Texas, United States, 78215 Contact: Eric J Lawitz, MD 210-253-3426 Principal Investigator: Eric Lawitz, MD United States, Virginia Liver Institute of Virginia-Bremo Recruiting Richmond, Virginia, United States, 23602 Principal Investigator: Mitchell Shiffman United States, Washington Swedish First Hill Campus Recruiting Seattle, Washington, United States, 98104 Principal Investigator: Kris Kowdley Collapse << |
| NCT00161083 | Cholecystolithiasis | Phase 4 | Completed | - | Netherlands ... more >> UMC Utrecht Utrecht, Netherlands, 3508 GA Collapse << |
| NCT01440309 | Primary Biliary Cirrhosis | Phase 1 | Unknown | December 2013 | China ... more >> Peking Union Medical College Hospital Recruiting Beijing, China Contact: Fengchun Zhang, MD zhangfccra@yahoo.com.cn Collapse << |
| NCT01899703 | Cholestasis, Intrahepatic | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Birmingham, West Midlands, United Kingdom, B15 2WB GSK Investigational Site Cambridge, United Kingdom GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT01430429 | Primary Biliary Cirrhosis | Phase 2 | Terminated | - | Italy ... more >> IRCCS Istituto Clinico Humanitas Rozzano, Italy, 20089 United Kingdom NIHR Liver Biomedical Research Unit, Queen Elizabeth Hospital Birmingham, United Kingdom, B15 2TH Collapse << |
| NCT00990639 | Alcoholic Liver Disease | Phase 1 Phase 2 | Completed | - | Korea, Republic of ... more >> Yonsei University Wonju College of Medicine Wonju Christian Hospital Wonju, Kangwon-do, Korea, Republic of, 220-701 Collapse << |
| NCT00004441 | Cystic Fibrosis | Not Applicable | Completed | - | United States, Colorado ... more >> Children's Hospital of Denver Denver, Colorado, United States, 80218 United States, Ohio Children's Hospital Medical Center - Cincinnati Cincinnati, Ohio, United States, 45229-3039 Italy University of Milan Milan, Italy, 20122 Collapse << |
| NCT03516006 | Primary Sclerosing Cholangitis | Phase 1 Phase 2 | Active, not recruiting | January 2024 | - |
| NCT02943707 | Liver Diseases | Phase 2 | Recruiting | October 2020 | China, Zhejiang ... more >> the First Affiliated Hospital of Wenzhou Medical University Recruiting Wenzhou, Zhejiang, China, 325000 Contact: yongping chen 8613505777281 13505777281@163.com Contact: lanman xu 8613587646315 13587646315@163.com Principal Investigator: yongping chen, MD Principal Investigator: lanman xu, PhD Principal Investigator: dazhi chen, MD Principal Investigator: minghua zheng, PhD Principal Investigator: keqing shi, MD Principal Investigator: yu huang, MD Principal Investigator: faling wu, MD Principal Investigator: ruicong chen, MD Principal Investigator: yunlei xiao, MD Collapse << |
| NCT00706381 | Healthy Volunteers | Not Applicable | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02916641 | Primary Biliary Cirrhosis | Phase 3 | Unknown | - | China, Shaanxi ... more >> Xijing Hosipital of Digestive Disease Recruiting Xi`an, Shaanxi, China, 710032 Contact: Ying Han, Ph.D 86-29-84771539 hanying@fmmu.edu.cn Contact: Yongquan Shi, Ph.D 86-29-84771515 shiyquan@fmmu.edu.cn Collapse << |
| NCT00004748 | Liver Cirrhosis, Biliary | Phase 3 | Completed | - | - |
| NCT00808743 | Familial Adenomatous Polyposis... more >> Duodenal Neoplasms Duodenal Polyps Collapse << | Phase 2 Phase 3 | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, Netherlands University Medical Center Groningen, Netherlands Leiden University Medical Center Leiden, Netherlands University Medical Center St. Radboud Nijmegen, Netherlands Erasmus Medical Center Rotterdam, Netherlands Collapse << |
| NCT02916290 | Primary Biliary Cirrhosis | Phase 3 | Unknown | - | China, Shaanxi ... more >> Xijing Hosipital of Digestive Disease Recruiting Xi`an, Shaanxi, China, 710032 Contact: Ying Han, Ph.D 86-29-84771539 hanying@fmmu.edu.cn Contact: Yongquan Shi, Ph.D 86-29-84771515 shiyquan@fmmu.edu.cn Collapse << |
| NCT02823366 | Primary Biliary Cirrhosis | Phase 3 | Unknown | - | China, Shaanxi ... more >> Xijing Hosipital Recruiting Xi`an, Shaanxi, China, 710032 Contact: Ying Han, Ph.D 86-29-84771539 hanying@fmmu.edu.cn Contact: Yongquan Shi, Ph.D 86-29-84771515 shiyquan@fmmu.edu.cn Collapse << |
| NCT02609048 | Primary Biliary Cirrhosis (PBC... more >>) Collapse << | Phase 2 | Terminated(Study stopped early... more >> after review of safety and efficacy demonstrated efficacy proof-of-concept and need for dose reduction.) Collapse << | - | - |
| NCT03476993 | Liver Cirrhosis, Biliary | Phase 2 | Recruiting | December 2021 | Russian Federation ... more >> State Budgetary Higher Vocational Education Institution I.M. Sechenov First Moscow State Medical University Recruiting Moscow, Russian Federation North-Western State Medical University named after I.I. Mechnikov Not yet recruiting Saint Petersburg, Russian Federation Smolensk state medical university Recruiting Smolensk, Russian Federation Collapse << |
| NCT00858858 | - | - | Completed | - | - |
| NCT00858858 | Gastroesophageal Reflux Diseas... more >>e Collapse << | Not Applicable | Completed | - | United States, Texas ... more >> VA North Texas Health Care System Dallas VA Medical Center, Dallas, TX Dallas, Texas, United States, 75216 Collapse << |
| NCT02955602 | Primary Biliary Cirrhosis | Phase 2 | Active, not recruiting | January 2020 | - |
| NCT00042549 | Cholelithiasis | Phase 4 | Terminated | - | United States, Texas ... more >> The Methodist Hospital Houston, Texas, United States, 77030 United States, Virginia Virgina Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT02078882 | Primary Biliary Cirrhosis | Phase 4 | Completed | - | United States, California ... more >> Univeristy of California Davis Medical Center Sacramento, California, United States, 95817 Collapse << |
| NCT03664596 | Non-alcoholic Steatohepatitis | Not Applicable | Recruiting | June 2019 | Kazakhstan ... more >> Medical Centre Hospital of the President's Affairs Administration Recruiting Astana, Kazakhstan, 010000 Contact: Bakytzhan Bimbetov, MD, PhD +7701 383 6072 bimbetov2010@mail.ru Sub-Investigator: Abay Zhangabylov, MD, PhD Sub-Investigator: Marzhan Rakhimzhanova, MD, MPH Collapse << |
| NCT02340247 | Severe Obesity | Phase 4 | Completed | - | Denmark ... more >> Hvidovre University Hospital Hvidovre, Denmark, DK-2650 Collapse << |
| NCT00006168 | Liver Cirrhosis, Biliary | Phase 3 | Completed | - | United States, California ... more >> Keck School of Medicine at U.S.C. Los Angeles, California, United States, 90033 U California Medical Center San Francisco, California, United States, 94143 United States, Connecticut Yale University School of Medicine New Haven, Connecticut, United States, 06520-8019 United States, Georgia Emory University School of Medicine Atlanta, Georgia, United States, 30322 United States, Missouri Saint Louis University St. Louis, Missouri, United States, 63104 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-3285 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Oregon Oregon Health Sciences University Portland, Oregon, United States, 97201 United States, Pennsylvania Albert Einstein Medical Center Philadelphia, Pennsylvania, United States, 19141 United States, Texas UT Southwestern Medical Center at Dallas Dallas, Texas, United States, 75235-9151 United States, Virginia Medical College of Virginia Richmond, Virginia, United States, 23298-0711 United States, Washington University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT02965911 | Primary Biliary Cirrhosis | Phase 1 Phase 2 | Recruiting | January 2019 | China ... more >> Beijing 302 hospital Recruiting Beijing, China, 100039 Contact: Zhengsheng Zou, Dr. Principal Investigator: Zhengsheng Zou, Dr. Collapse << |
| NCT01456468 | Cholangitis, Sclerosing | Phase 1 | Completed | - | United States, Connecticut ... more >> Yale University School of Medicine - 333 Cedar St - 1080 LMP New Haven, Connecticut, United States, 06520 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00857480 | Hepatic Cirrhosis | Phase 1 | Completed | - | Bulgaria ... more >> MHAPT "Zarita Johanna" University Hospital Sofia, Bulgaria, 1431 Collapse << |
| NCT03665519 | Primary Biliary Cirrhosis | Not Applicable | Recruiting | December 2020 | Kazakhstan ... more >> National Research Medical Center Recruiting Astana, Kazakhstan, 010000 Contact: Galiya Shaimardanova, PhD +77017299148 galiya_masugut@mail.ru Contact: Gulmira Dossatayeva, MD, MBA +77015127500 g_dossatayeva@inbox.ru Principal Investigator: Abay Baigenzhin, MD, PhD Sub-Investigator: Gulmira Dossatayeva, MD, MBA Sub-Investigator: Maiya Zhumabayeva, MD Sub-Investigator: Galiya Shaimardanova, PhD Collapse << |
| NCT02622685 | Healthy | Phase 1 | Completed | - | - |
| NCT01141296 | Primary Biliary Cirrhosis | Phase 2 | Withdrawn(Lack of funding) | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 United States, Minnesota Mayo Clinic Rochester Rochester, Minnesota, United States, 55902 Collapse << |
| NCT01073202 | Liver Transplantation ... more >> Ischemia-reperfusion Injury Cholestasis Collapse << | Not Applicable | Completed | - | China, Shanghai ... more >> Shanghai First People's Hospital Shanghai, Shanghai, China, 200080 Collapse << |
| NCT03678480 | Primary Sclerosing Cholangitis... more >> Cholangitis Cholangitis, Sclerosing Bile Duct Diseases Biliary Tract Diseases Digestive System Diseases Adolescent Collapse << | Phase 2 | Not yet recruiting | November 4, 2019 | - |
| NCT03201952 | Glucose Intolerance ... more >> Gastric Bypass Surgery Collapse << | Phase 1 | Recruiting | July 1, 2019 | United States, Tennessee ... more >> Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Contact: Michael Dole, MD 864-616-1389 michael.dole@vanderbilt.edu Collapse << |
| NCT01829698 | Cholestatic Liver Disease | Phase 3 | Completed | - | China, Beijing ... more >> Liver Research Center,Beijing Friendship Hospital Beijing, Beijing, China, 100050 Collapse << |
| NCT03124108 | Primary Biliary Cholangitis (P... more >>BC) Collapse << | Phase 2 | Recruiting | April 2018 | United States, Arizona ... more >> Mayo Clinic in Arizona Recruiting Phoenix, Arizona, United States, 85054 Contact: Elizabeth J. Carey, MD 480-342-2000 United States, Florida Schiff Center for Liver Diseases Recruiting Miami, Florida, United States, 33136 Contact: Cynthia Levy, MD 305-243-4615 United States, Massachusetts Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Daniel Pratt, MD 617-724-3836 Beth Israel Deaconess Medical Center Recruiting Boston, Massachusetts, United States, 02215 Contact: Alan Bonder, MD 617-632-1086 United States, New York Northwell Health - Sandra Atlas Bass Center for Liver Disease Recruiting Manhasset, New York, United States, 11030 Contact: Henry C. Bodenheimer, MD 516-562-4664 United States, Texas University of Texas Southwestern Medical Center Recruiting Dallas, Texas, United States, 75390 Contact: Marlyn Mayo, MD 214-648-2725 Baylor College of Medicine - Advanced Liver Therapies Recruiting Houston, Texas, United States, 77030 Contact: John M. Vierling, MD 832-355-8966 United States, Virginia University of Virginia Recruiting Charlottesville, Virginia, United States, 22908 Contact: Stephen H. Caldwell, MD 434-924-2626 Virginia Commonwealth University Recruiting Richmond, Virginia, United States, 23298 Contact: Velimir Luketic, MD 804-828-4060 United States, Washington Swedish Organ Transplant and Liver Center Recruiting Seattle, Washington, United States, 98104 Contact: Kris Kowdley, MD 206-386-3880 Germany Clinic for Gastroenterology and Hepatology Recruiting Köln, Germany, 50937 Contact: Münevver Demir, MD +49 (0)2214 787334 Johannes Gutenberg University Recruiting Mainz, Germany, 55131 Contact: Jorn M Schattenberg, MD +49 (0)6131 176831 Spain Hospital de la Santa Creu i Sant Pau Recruiting Barcelona, Spain, 08025 Contact: Adolfo Gallego Moya, MD +34 932 919-000 Liver Unit, University of Barcelona Recruiting Barcelona, Spain, 08036 Contact: Albert Pares, MD +34 932 275-753 United Kingdom Queen Elizabeth Hospital Recruiting Birmingham, United Kingdom, B15 2GW Contact: Gideon Hirschfield, MD +44 (0)121 415-8700 Addenbrooke's Hospital Recruiting Cambridge, United Kingdom, CB2 0QQ Contact: George Mells, MD +44 (0)122 324-5151 The Royal Liverpool University Hospital Recruiting Liverpool, United Kingdom, L7 8XP Contact: Imran Patanwala, MD +44 (0)151 706-2000 Royal Free London NHS Foundation Trust Not yet recruiting London, United Kingdom, NW3 2QG Contact: Douglas Thorburn, MD +44 (0)207 794-0500 Institute of Liver Studies, King's College Hospital Recruiting London, United Kingdom, SE5 9RS Contact: Michael Heneghan, MD +44 (0)203 299-7615 Royal Victoria Infirmary Recruiting Newcastle upon Tyne, United Kingdom, NE1 4LP Contact: David Jones, MD +44 (0)191 222-8335 Collapse << |
| NCT01389973 | Primary Biliary Cirrhosis | Phase 2 | Completed | - | United States, Florida ... more >> Jacksonville, Florida, United States Miami, Florida, United States Naples, Florida, United States United States, Mississippi Tupelo, Mississippi, United States United States, New York New York, New York, United States United States, Texas Houston, Texas, United States Canada, British Columbia Vancouver, British Columbia, Canada Canada, Nova Scotia Halifax, Nova Scotia, Canada Canada, Ontario Toronto, Ontario, Canada Canada, Quebec Montreal, Quebec, Canada Collapse << |
| NCT01389973 | - | - | Completed | - | - |
| NCT03659058 | Fibrosis, Liver | Not Applicable | Completed | - | - |
| NCT03226067 | Primary Biliary Cirrhosis | Phase 2 | Active, not recruiting | September 26, 2019 | - |
| NCT02823353 | Primary Biliary Cirrhosis | Phase 3 | Unknown | July 2018 | China, Shaanxi ... more >> Xijing Hosipital Recruiting Xi`an, Shaanxi, China, 710032 Contact: Ying Han, MD,PH.D 8602984771539 hanying@fmmu.edu.cn Collapse << |
| NCT03586674 | Chronic Cholestasis | Phase 2 | Completed | - | Egypt ... more >> Dr. Yassin Abdel Ghaffar Charity Center For Liver Diseases & Researches Cairo, Nasr City, Egypt National liver istitute Shibīn Al Kawm, Egypt Collapse << |
| NCT03322943 | - | - | Recruiting | December 1, 2018 | China, Sichuan ... more >> West China Hospital Recruiting Chengdu, Sichuan, China, 610041 Contact: Li Yang, MD +8618980601276 yangli_hx@scu.edu.cn Collapse << |
| NCT01904058 | PBC Primary B... more >>iliary Cirrhosis Collapse << | Phase 2 | Completed | - | - |
| NCT01654731 | PBC | Phase 3 | Completed | - | France ... more >> Hepatology department - Hopital Saint Antoine Paris, France, 75012 Collapse << |
| NCT00004784 | Liver Cirrhosis, Biliary | Phase 3 | Completed | - | - |
| NCT03602560 | Primary Biliary Cholangitis | Phase 3 | Recruiting | December 2021 | United States, Arizona ... more >> Institute for Liver Health Recruiting Chandler, Arizona, United States, 85224 Contact: Dana Robledo 480-686-8874 drobledo@iliverhealth.org Principal Investigator: Anita Kohli, MD United States, Colorado Univeristy of Colorado Denver and Hospital Recruiting Aurora, Colorado, United States, 80045 Contact: Joselin Gonzalez-Diaz 303-724-1866 joselin.gonzalez-diaz@ucdenver.edu Principal Investigator: Lisa Forman, MD United States, Florida Excel Medical Clinical Trials, LLC Recruiting Boca Raton, Florida, United States, 33434 Contact: Jennifer Olivar 561-756-8206 jolivar@emclinicaltrials.com Principal Investigator: Seth J Baum, MD Florida Reserach Institute Recruiting Lakewood Ranch, Florida, United States, 34211 Contact: Kalina Miller 941-727-7772 kalina.miller@fdhs.com Principal Investigator: Guy W Neff, MD United States, Georgia Digestive Healthcare of Georgia Recruiting Atlanta, Georgia, United States, 30309 Contact: Debeh Legaspi 678-686-5073 dlegaspi@digestivehealthcare.net Principal Investigator: Michael R Galambos, MD United States, Indiana Indianapolis Gastroenterology Research Foundation Recruiting Indianapolis, Indiana, United States, 46237 Contact: Sheila Thompson 317-865-2959 sthompson@indygastro.com Principal Investigator: David C Pound, MD United States, Minnesota Minnesota Gastroenterlogy, P.A. Recruiting Maplewood, Minnesota, United States, 55117 Contact: Gavin Mark 612-870-5597 gavin.mark@mngastro.com Principal Investigator: Donald L Zogg, MD United States, Mississippi Southern Therapy and Advance Research (STAR) LLC Recruiting Jackson, Mississippi, United States, 39216 Contact: Evelyn Arendale 769-251-5674 earendale@jacksonliverandgi.com Principal Investigator: Brian B Borg, MD United States, Missouri Kansas City Research Institute Recruiting Kansas City, Missouri, United States, 64131 Contact: Kelly Vargas 816-759-5274 kvargas@kcresearchinstitute.com Contact: Jeanne Zirkle (816) 759-5274 jzirkle@kcresearchinstitute.com Principal Investigator: Bradley L Freilich, MD United States, New York Concorde Medical Group Recruiting New York, New York, United States, 10016 Contact: Resmi Varughese 212-726-9472 rvarughese@concordemed.com Principal Investigator: Hillel Tobias, MD NYU Langone Health Recruiting New York, New York, United States, 10016 Contact: Kristyn Pierce 646-501-8468 Kristyn.Pierce@nyulangone.org Principal Investigator: Carmen Stanca, MD University of Rochester Medical Center Recruiting Rochester, New York, United States, 14642 Contact: Juliana Posato 585-275-4711 juliana_posato@urmc.rochester.edu Principal Investigator: Jonathan C Huang, DO United States, North Carolina Asheville Gastroenterology Associates, a Division of Digestive Health Partners, P.A. Recruiting Asheville, North Carolina, United States, 28801 Contact: Amber Bartlett 828-254-0881 abartlett@ncdhp.com Principal Investigator: Adam S Zivony, MD United States, Pennsylvania Northeast Clinical Research Center, LLC Recruiting Bethlehem, Pennsylvania, United States, 18017 Contact: Jennifer Jones 610-433-4100 jennifer.jones@necresearch.org Principal Investigator: Adam Peyton, DO United States, Tennessee Gastro One Recruiting Germantown, Tennessee, United States, 38138 Contact: Keosha Henderson 901-682-6540 khenderson@gastro1.com Principal Investigator: Ziad Younes, MD GIA Clinical Trials, LLC Recruiting Knoxville, Tennessee, United States, 37909 Contact: Kathy Karnes 865-558-0687 kkarnes@gihealthcare.com Principal Investigator: Jason Huffman, MD United States, Texas Texas Digestive Disease Consultants Recruiting Fort Worth, Texas, United States, 76104 Contact: Neal Bhakta 817-310-4478 nbhakta@tddctx.com Principal Investigator: Apurva A Modi, MD Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Jignesh Patel 713-798-5242 jignesh.patel@bcm.edu Principal Investigator: John M Vierling, MD American Research Corporation at Texas Liver Institute Recruiting San Antonio, Texas, United States, 78215 Contact: MaryLou Hernandez 210-253-3426 mhernandez@txliver.com Principal Investigator: Eric J Lawitz, MD Pinnacle Clinical Research Recruiting San Antonio, Texas, United States, 78229 Contact: Kathy Marotta 210-982-0320 ext 1437 kmarotta@pinnacleresearch.com Principal Investigator: Stephen Harrison, MD United States, Virginia Bon Secours Richmond Community Hospital Recruiting Richmond, Virginia, United States, 23226 Contact: Susan Vollum 804-977-8921 susan_vollum@bshsi.org Principal Investigator: Mitchell L Shiffman, MD Collapse << |
| NCT01904058 | - | - | Completed | - | - |
| NCT01974336 | Short Bowel Syndrome | Phase 4 | Completed | - | China, Jiangsu ... more >> Jinling Hospital Nanjing, Jiangsu, China, 200002 Collapse << |
| NCT00285597 | Heart Failure, Congestive | Phase 2 | Unknown | - | United Kingdom ... more >> National Heart and Lung Institute London, United Kingdom, SW3 6LY Collapse << |
| NCT03000218 | Overweight Ab... more >>normal Liver Function Tests Collapse << | Phase 1 | Unknown | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seoul, Korea, Republic of Contact: Jae-Yong Chung, MD, PhD +82-031-787-3955 Principal Investigator: Jae-Yong Chung, MD, PhD Collapse << |
| NCT03092765 | Primary Biliary Cholangitis | Phase 2 | Active, not recruiting | October 2018 | Japan ... more >> EA Pharma trial site Nagoya, Aichi, Japan EA Pharma trial site Matsudo, Chiba, Japan EA Pharma trial site Touon, Ehime, Japan EA Pharma trial site Yoshida, Fukui, Japan EA Pharma trial site Kurume, Fukuoka, Japan EA Pharma trial site Maebashi, Gunma, Japan EA Pharma trial site #1 Sapporo, Hokkaido, Japan EA Pharma trial site #2 Sapporo, Hokkaido, Japan EA Pharma trial site Kobe, Hyogo, Japan EA Pharma trial site Nishinomiya, Hyogo, Japan EA Pharma trial site Inashiki, Ibaraki, Japan EA Pharma trial site Morioka, Iwate, Japan EA Pharma trial site Kita, Kagawa, Japan EA Pharma trial site Takamatsu, Kagawa, Japan EA Pharma trial site #1 Yokohama, Kanagawa, Japan EA Pharma trial site #2 Yokohama, Kanagawa, Japan EA Pharma trial site Sendai, Miyagi, Japan EA Pharma trial site Matsumoto, Nagano, Japan EA Pharma trial site Oomura, Nagasaki, Japan EA Pharma trial site Kashihara, Nara, Japan EA Pharma trial site Nakagami, Okinawa, Japan EA Pharma trial site Hirakata, Osaka, Japan EA Pharma trial site #1 Suita, Osaka, Japan EA Pharma trial site #2 Suita, Osaka, Japan EA Pharma trial site Ageo, Saitama, Japan EA Pharma trial site Iruma, Saitama, Japan EA Pharma trial site Shimotsuga, Tochigi, Japan EA Pharma trial site Bunkyo, Tokyo, Japan EA Pharma trial site Itabashi, Tokyo, Japan EA Pharma trial site #1 Minato, Tokyo, Japan EA Pharma trial site #2 Minato, Tokyo, Japan EA Pharma trial site Musashino, Tokyo, Japan EA Pharma trial site Shinjuku, Tokyo, Japan EA Pharma trial site #1 Fukuoka, Japan EA Pharma trial site #2 Fukuoka, Japan EA Pharma trial site Fukushima, Japan EA Pharma trial site #1 Hiroshima, Japan EA Pharma trial site #2 Hiroshima, Japan EA Pharma trial site Kagoshima, Japan EA Pharma trial site Kumamoto, Japan EA Pharma trial site Kyoto, Japan EA Pharma trial site Niigata, Japan EA Pharma trial site Okayama, Japan EA Pharma trial site Osaka, Japan EA Pharma trial site Saga, Japan EA Pharma trial site Yamagata, Japan Collapse << |
| NCT00550862 | Liver Cirrhosis, Biliary | Phase 2 | Completed | - | - |
| NCT00746486 | Primary Biliary Cirrhosis | Phase 3 | Terminated(Study stopped based... more >> on the recommendation of the IDMC after a planned Interim Analysis) Collapse << | September 2017 | France ... more >> Hôpital Saint-Antoine Paris, France, 75571 Germany Universitätsklinikum Bonn Bonn, NRW, Germany, 53105 Collapse << |
| NCT02967250 | Parkinson Disease | Phase 1 | Recruiting | March 1, 2019 | United States, Minnesota ... more >> Center for Magnetic Resonance Research Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Lisa Coles 410-926-4788 durh0016@umn.edu Principal Investigator: Lisa Coles, MS, PhD Sub-Investigator: Paul Tuite, MD Collapse << |
| NCT02385032 | Healthy | Phase 1 | Completed | - | - |
| NCT00004315 | Cystic Fibrosis ... more >> Gastrointestinal Diseases Cholestasis Collapse << | Phase 2 | Unknown | - | United States, Ohio ... more >> Children's Hospital Medical Center - Cincinnati Cincinnati, Ohio, United States, 45229-3039 Collapse << |
| NCT01226823 | Intrahepatic Cholestasis of Pr... more >>egnancy Collapse << | Phase 3 | Withdrawn(Modified insurance r... more >>equests by a new law released after its approval.) Collapse << | November 2013 | Italy ... more >> Divisione di Gastroenterologia, IRCCS Casa Sollievo della Sofferenza San Giovanni Rotondo, Foggia, Italy, 71013 UOC Ostetricia e Ginecologia, Ospedale Maggiore Bologna, Italy, 40100 S.Orsola-Malpighi Hospital Bologna, Italy, 40138 Gastroenterology and Liver Clinic, Azienda Ospedaliero-Universitaria, University of Modena and Reggio Emilia Modena, Italy Dept. of Surgical and Gastroenterological Sciences, University of Padova Padua, Italy, 34100 Gastroenterology Unit, Policlinic of Palermo Palermo, Italy Internal Medicine Department, Gemelli Hospital, Catholic University of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT00550862 | - | - | Completed | - | - |
| NCT01857284 | Primary Biliary Cirrhosis | Phase 3 | Completed | - | China, Beijing ... more >> Beijing 302 Hospital Beijing, Beijing, China Beijing Ditan Hospital Beijing, Beijing, China Beijing Friendship Hospital Beijing, Beijing, China Beijing Youan Hospital Beijing, Beijing, China Chinese PLA General Hospital Beijing, Beijing, China Peiking University First Hosptial Beijing, Beijing, China Peking University People's Hospital Beijing, Beijing, China China, Guangdong First Affiliated Hospital,SunYat-Sen University Guangzhou, Guangdong, China Nanfang Hospital of Southern Medical University Guangzhou, Guangdong, China Third Affiliated Hospital,SunYat-Sen University Guangzhou, Guangdong, China China, Hunan Tongji Hospital Wuhan, Hunan, China China, Shanghai 85 Military Hospital Shanghai, Shanghai, China Eastern Hepatobiliary Surgery Hospital Shanghai, Shanghai, China Huashan Hospital Shanghai, Shanghai, China NO.3 People's Hospital Affiliated to Shanghai Jiao Tong University School of Medicine Shanghai, Shanghai, China RenJi Hospital Shanghai, Shanghai, China RuiJin Hospital Shanghai, Shanghai, China ShangHai Changzheng Hospital Shanghai, Shanghai, China Shanghai Public Health Clinical Center Shanghai, Shanghai, China Shanghai Zhongshan Hospital Shanghai, Shanghai, China China, Shanxi Xijing Hospital Xian, Shanxi, China China, Sichuan West China Hospital Chengdu, Sichuan, China China, Yunnan First Affiliated Hospital Of KunMing Medical College Kunming, Yunnan, China China, Zhejiang The Sixth People's Hospital of Hangzhou Hangzhou, Zhejiang, China First Affiliated Hospital of Zhejiang University Zhejiang, Zhejiang, China Collapse << |
| NCT01088607 | Primary Sclerosing Cholangitis | Phase 1 | Completed | - | United States, Arizona ... more >> Phoenix Children's Hospital Phoenix, Arizona, United States, 85016 United States, California Children's Hospital Los Angeles Los Angeles, California, United States, 90027 University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado Children's Hospital Colorado Aurora, Colorado, United States, 80045 United States, Connecticut Yale New Haven Children's Hospital New Haven, Connecticut, United States, 06510 United States, Georgia Children's Healthcare of Atlanta, Emory University School of Medicine Atlanta, Georgia, United States, 30322 United States, Illinois Northwestern University Chicago, Illinois, United States, 60614 United States, New York Mount Sinai School of Medicine New York, New York, United States, 07624 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Tennessee University of Tennessee Health Science Center Memphis, Tennessee, United States, 38103 United States, Texas Texas Children's Hospital Houston, Texas, United States, 77030 Collapse << |
| NCT03056274 | Intrahepatic Cholestases ... more >> Pregnancy Complications Collapse << | Phase 2 Phase 3 | Not yet recruiting | September 2019 | - |
| NCT02420496 | Cholestasis L... more >>iver Disease Collapse << | Phase 2 | Withdrawn | - | United States, Texas ... more >> University Hospital San Antonio, Texas, United States, 78229 Collapse << |
| NCT03004118 | Functional Dyspepsia | Phase 4 | Unknown | November 2017 | - |
| NCT01149304 | Colorectal Cancer ... more >> Liver Metastases Irradiation Damage Radiation Induced Liver Disease Collapse << | Phase 2 | Completed | - | Germany ... more >> Clinic for Radiology and Nuclear Medicine Magdeburg, Sachsen-Anhalt, Germany, 39120 Collapse << |
| NCT01141322 | Hepatitis, Toxic | Phase 4 | Unknown | July 2010 | Taiwan ... more >> Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 11217 Contact: Yi-Shin Huang, M.D. +886-2-2871-2121 ext 2015 yshuang@vghtpe.gov.tw Principal Investigator: Yi-Shin Huang, M.D. Collapse << |
| NCT00470171 | Serum Levels of ALAT Transamin... more >>ases Serum Markers for Fibrosis and Hepatic Inflammation Collapse << | Phase 2 | Completed | - | France ... more >> La Pitié Salpétrière Hospital Paris, France, 75013 Collapse << |
| NCT02841306 | Rhegmatogenous Retinal Detachm... more >>ent Collapse << | Phase 1 | Recruiting | December 2017 | Switzerland ... more >> University of Lausanne Recruiting Lausanne, Switzerland Contact: Alejandra Daruich, MD +41216268858 adaruich.matet@gmail.com Collapse << |
| NCT02472509 | Hemolytic Disorders ... more >> Gallstones Collapse << | Phase 4 | Terminated(Study not feasible ... more >>- very low recruitment rate) Collapse << | - | Israel ... more >> Shaare Zedek Medical Center Jerusalem, Israel, 91031 Collapse << |
| NCT03188146 | - | - | Recruiting | May 1, 2028 | China ... more >> Centre of non-infectious liver diseases, Beijing 302 Hospital, Beijing, China. Recruiting Beijing, China, 86 Contact: Ang Huang, MD duanzpyouan@gmail.com Principal Investigator: George Lau, MD Sub-Investigator: Ang Huang, MD Collapse << |
| NCT01337440 | Type 2 Diabetes Mellitus ... more >> Chronic Liver Disease Collapse << | Phase 4 | Unknown | March 2013 | Japan ... more >> Internal medicine, Kanazawa university hospital Recruiting Kanazawa, Ishikawa, Japan, 920-8641 Contact: Toshinari Takamura, MD, PhD +81-76-265-2233 ttakamura@m-kanazawa.jp Sub-Investigator: Kosuke R Shima, MD Collapse << |
| NCT01576458 | Intrahepatic Cholestasis ... more >> Pregnancy Collapse << | Phase 3 | Completed | - | Finland ... more >> Turku University Hospital Turku, Finland, 20520 Collapse << |
| NCT02016365 | Transthyretin Amyloidosis ... more >> Cardiomyopathy Collapse << | Phase 2 | Completed | - | Sweden ... more >> Dept of Clinical Medicin, Ptieå Hospital Piteå, Sweden, SE-941 28 Dept of clinical medicin, Skellefteå Hospital Skellefteå, Sweden, SE-931 86 Dept of Clinical Medicine, Umeå University Hospital Umeå, Sweden, SE-90185 Collapse << |
| NCT02936596 | Hepatitis, Autoimmune ... more >> Cholangitis Liver Cirrhosis, Biliary Cholestasis Collapse << | Not Applicable | Recruiting | August 2018 | China, Sichuan ... more >> West China Hospital Recruiting Chengdu, Sichuan, China, 610041 Contact: Xiaoli Fan, MM +8618008028017 13980433451@163.com Contact: Li Yang, MD yangli_hx@scu.edu.cn Collapse << |
| NCT02937012 | Primary Biliary Cirrhosis | Phase 3 | Recruiting | December 2019 | Mexico ... more >> Instituto Nacional de Ciencias Medicas y Nutricion Salvador Zubiran Recruiting Mexico City, Mexico, 14000 Contact: Edgardo Eric Lopez Mendez, MD (01)(55) 54870900 ext 2710 ericlopezmendez@yahoo.com.mx Contact: Sergio Gabriel Munoz Martinez, MD (01)(55) 54870900 ext 2710 sergio_sg@hotmail.com Principal Investigator: Edgardo Eric Lopez Martinez, MD Sub-Investigator: Sergio Gabriel Munoz Martinez, MD Sub-Investigator: Ignacio Garcia Juarez, MD Sub-Investigator: Ernesto Marquez Guillen, MD Sub-Investigator: Carlos Moctezuma Velazquez, MD Sub-Investigator: Alejandra Tepox Padron, MD Collapse << |
| NCT00200343 | Chronic Hepatitis C | Phase 3 | Completed | - | Japan ... more >> Department of Gastroenterology, University of Tokyo hongo, bunkyo-ku, Tokyo, Japan Collapse << |
| NCT00134758 | Adenomatous Polyposis Coli, Fa... more >>milial Collapse << | Phase 2 Phase 3 | Unknown | October 2009 | France ... more >> Saint-Antoine Hospital Paris, France, 75012 Collapse << |
| NCT00062023 | Colorectal Cancer | Phase 2 | Terminated(Study closed by the... more >> NCI.) Collapse << | - | United States, Texas ... more >> M.D. Anderson Cancer Center at University of Texas Houston, Texas, United States, 77030-4009 Collapse << |
| NCT02338635 | End-Stage Kidney Disease | Phase 4 | Unknown | January 2018 | - |
| NCT03602976 | Hepatic Sarcoidosis, Elevated ... more >>Alkaline Phosphatase Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Pennsylvania ... more >> University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Kelly A Borges 215-615-3755 kelly.borges@uphs.upenn.edu Principal Investigator: Ethan M Weinberg, MD, MS Principal Investigator: K. Rajender Reddy, MD Collapse << |
| NCT01548079 | Non-alcoholic Fatty Liver Dise... more >>ase Morbid Obesity Collapse << | Not Applicable | Completed | - | - |
| NCT00873275 | Colorectal Cancer | Phase 1 | Active, not recruiting | September 2019 | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010-3000 Collapse << |
| NCT02021110 | Polycystic Liver Disease (PLD)... more >>: Polycystic Kidney, Autosomal Dominant Polycystic Liver Disease Collapse << | Phase 2 | Completed | - | Netherlands ... more >> Radboud University Medical Centre Nijmegen Nijmegen, Gelderland, Netherlands Academic Medical Centre Amsterdam Amsterdam, Netherlands Spain Donostia University Hospital San Sebastian, Spain Collapse << |
| NCT02789644 | Aged | Phase 1 | Unknown | July 2016 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seoul, Korea, Republic of Contact: Jae-Yong Chung, MD, PhD +82-031-787-3955 Principal Investigator: Jae-Yong Chung, MD, PhD Collapse << |
| NCT00200343 | - | - | Completed | - | - |
| NCT03724175 | Ulcerative Colitis ... more >> Pouchitis Collapse << | Phase 3 | Not yet recruiting | December 2050 | - |
| NCT00059202 | Sclerosing Cholangitis | Phase 2 Phase 3 | Completed | - | United States, Arizona ... more >> Mayo Clinic Scottsdale, Arizona, United States United States, Florida Mayo Clinic Jacksonville, Florida, United States United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, Missouri St. Louis University St. Louis, Missouri, United States United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States United States, Virginia Medical College of Virginia Richmond, Virginia, United States United States, Washington University of Washington Seattle, Washington, United States Collapse << |
| NCT02033876 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00514774 | Huntington Disease | Phase 1 | Unknown | June 2009 | United States, Oregon ... more >> Oregon Health & Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT01510860 | Primary Biliary Cirrhosis | Phase 4 | Completed | - | Germany ... more >> Klinikum Grosshadern Munich, Germany, 81377 Collapse << |
| NCT03489889 | Ursodeoxycholic Acid ... more >> Primary Biliary Cirrhosis Collapse << | Not Applicable | Completed | - | Brazil ... more >> Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo São Paulo, Brazil, 05403-000 Collapse << |
| NCT02319629 | Morbid Obesity | Not Applicable | Recruiting | December 2018 | Israel ... more >> surgery department A Recruiting Afula, Israel Contact: Nasser Sakran, MD Collapse << |
| NCT01097304 | Barrett Esophagus ... more >> Esophageal Carcinoma Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Southern Arizona Veterans Affairs Health Center Tucson, Arizona, United States, 85723 Arizona Cancer Center-North Campus Tucson, Arizona, United States, 85724-5024 University of Arizona Health Sciences Center Tucson, Arizona, United States, 85724 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT00909610 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Pharma Medica Research Inc. Toronto, Ontario, Canada, M1R 5A3 Collapse << |
| NCT00909610 | - | - | Completed | - | - |
| NCT00846963 | Cholestasis | Phase 2 Phase 3 | Completed | - | Canada, Quebec ... more >> CHU Sainte-Justine Montréal, Quebec, Canada, H3T 1C5 Collapse << |
| NCT01688375 | Obstructive Jaundice | Phase 2 | Unknown | December 2013 | Serbia ... more >> University clinical centre of Kosovo, Department of general surgery Not yet recruiting Pristina, Kosovo, Serbia, 10000 Contact: Enver H Fekaj, PhD student 37744279296 enverfekaj@hotmail.com Principal Investigator: Enver H Fekaj, PhD student University clinical centre of Kosovo Pristina, Kosovo, Serbia, 10000 University clinical centre of Kosovo Not yet recruiting Pristina, Kosovo, Serbia, 10000 Contact: Enver H Fekaj, PhD student 37744279296 enverfekaj@hotmail.com Collapse << |
| NCT00909753 | - | - | Completed | - | - |
| NCT03512808 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Pharma medica research Inc. Mississauga, Ontario, Canada, 6100 Collapse << |
| NCT01097304 | - | - | Completed | - | - |
| NCT03512821 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Pharma medica research Inc. Mississauga, Ontario, Canada, 6100 Collapse << |
| NCT02244944 | - | - | Terminated(Failure to recruit) | - | - |
| NCT00909753 | Healthy | Phase 1 | Completed | - | Canada, Ontario ... more >> Pharma Medica Research Inc. Toronto, Ontario, Canada, M1R 5A3 Collapse << |
| NCT02748616 | Diarrhea | Phase 4 | Recruiting | November 2020 | United States, New York ... more >> New York University Langone Medical Center Recruiting New York, New York, United States, 10016 Contact: Lea Chen lea.chen@nyumc.org Principal Investigator: Norman Javitt, MD Collapse << |
| NCT02244944 | Nonalcoholic Fatty Liver Disea... more >>se (NAFLD) Collapse << | Phase 2 | Terminated(Failure to recruit) | - | United States, Kentucky ... more >> University of Kentucky Medical Center Lexington, Kentucky, United States, 40536 Collapse << |
| NCT02721862 | Cholelithiasis | Phase 3 | Recruiting | March 2019 | Lebanon ... more >> American University of Beirut Medical Center Recruiting Beirut, Lebanon, 1107 2020 Contact: Ramzi S. Alami, MD, FACS +961-1-350000 ext 5238 ra204@aub.edu.lb Makassed General Hospital Recruiting Beirut, Lebanon Contact: Hayssam Fawal, MD, FACS +961-3-824228 hayssamfawal@gmail.com Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
